This content is sponsored by AstraZeneca.
This content is intended only for US Healthcare Providers.

IMFINZI plus carboplatin and paclitaxel (CP) brings another treatment option to patients with primary advanced or recurrent endometrial cancer that is mismatch repair deficient


As of June 2024, the FDA approved IMFINZI® (durvalumab) in combination with carboplatin and paclitaxel (CP) followed by IMFINZI as a single agent for the treatment of adult patients with primary advanced or recurrent endometrial cancer that is mismatch repair deficient (dMMR).
IMFINZI + CP followed by IMFINZI as a single agent led to a
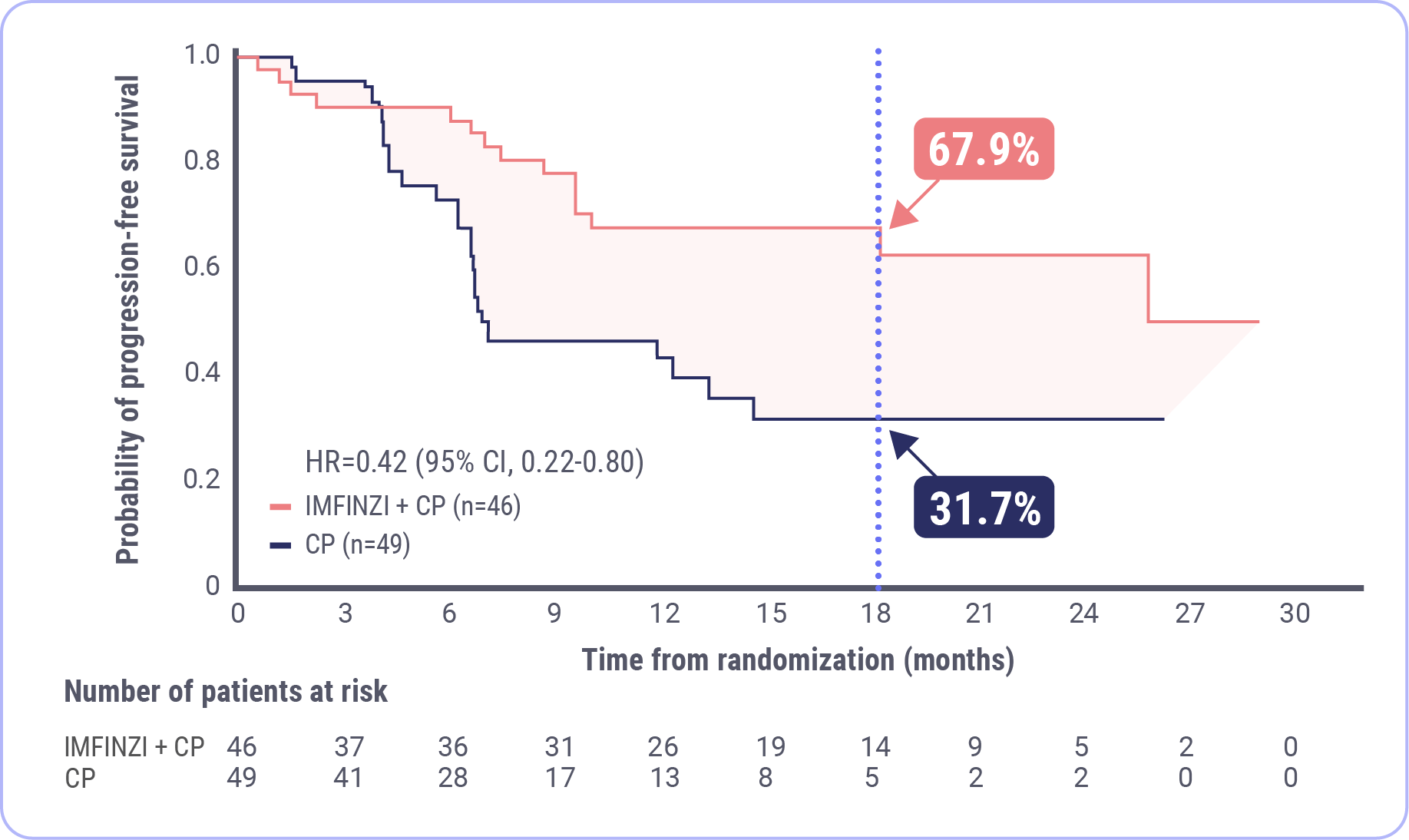
in the risk of progression or death in patients with dMMR endometrial cancer versus chemotherapy alone (HR 0.42; 95% CI, 0.22-0.80).
Median PFS was NR with IMFINZI + CP
(95% CI, NR-NR)
Median PFS was 7.0 months with CP
(95% CI, 6.7-14.8)
FDA approval was based on a prespecified dMMR subgroup (n=95). The prespecified PFS subgroup analysis was exploratory and not designed to assess a statistical difference between treatment groups.
CI=confidence interval; CP=carboplatin and paclitaxel; dMMR=mismatch repair deficient; FDA=United States Food and Drug Administration; HR=hazard ratio; NR=not reached; PFS=progression-free survival.
Unmet Need and Recent Advancements
Endometrial cancer (EC) is the most common gynecologic malignancy in the US, with rising incidence and mortality

In the US:
EC is the 4th most common cancer in women
EC accounts for 6% of cancers in women
Approximately 69,120 new cases of EC will be diagnosed in 2025
Approximately 13,860 women will die from EC in 2025
The number of new cases will double to 122,000 cases per year by 2030 if current trends continue
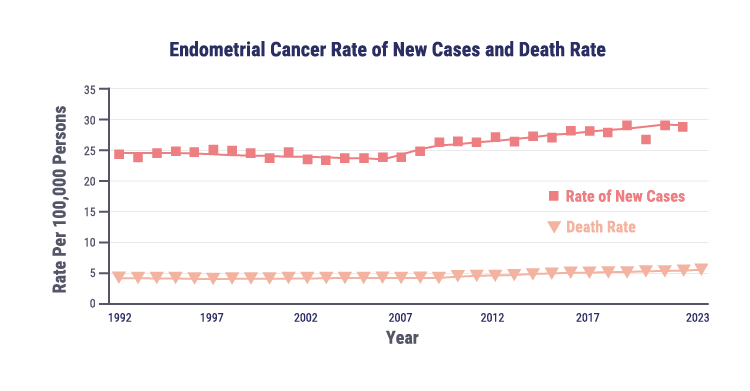
The rising incidence of endometrial cancer may be associated with several risk factors, including obesity, diabetes, and changes in hormone therapy use.
There is an unmet need for more treatment options

Considering the rising incidence and mortality, it is more important than ever to bring additional treatment options to patients with endometrial cancer.
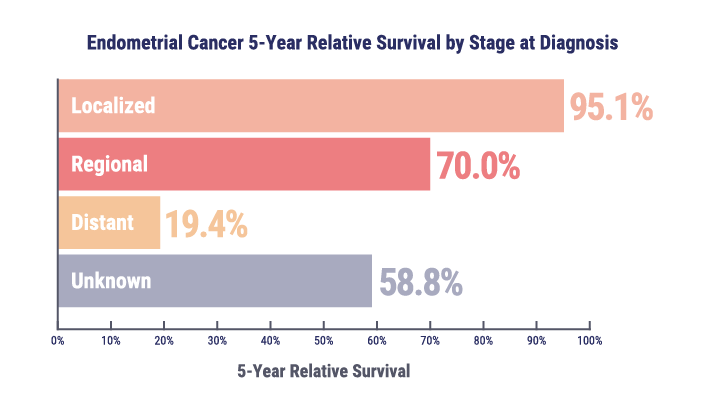
Patients diagnosed with localized disease have a 5-year survival rate of approximately 95%, whereas the survival rate falls below 20% for those with metastatic or recurrent disease.
Approximately 18% of women with endometrial cancer experience disease recurrence.
There is a need for additional treatment options for patients with primary advanced or recurrent dMMR endometrial cancer.
The endometrial cancer treatment landscape is shifting

Historically, standard of care for advanced or recurrent endometrial cancer has been platinum-based chemotherapy with carboplatin plus paclitaxel.
However, outcomes for patients with advanced or recurrent endometrial cancer remain poor when treated with conventional chemotherapy regardless of mismatch repair (MMR) status, with 5-year survival rates below 20%.

Mismatch repair status is an established prognostic biomarker

Testing patients for mismatch repair (MMR) status is recommended soon after diagnosis, as MMR status dictates if patients are eligible to receive specific precision medicine treatments.
Patients can be deficient (dMMR) or proficient (pMMR) in the MMR pathway that repairs DNA damage. The MMR pathway corrects base mismatches and insertion or deletion mismatches that can occur during DNA replication, and therefore plays an important role in maintaining genomic stability. Defects in MMR increase mutation rates and can be oncogenic. dMMR tumors are more likely to produce abnormal proteins that attract immune cells and are generally more responsive to immunotherapy (IO) treatment, in comparison to pMMR tumors.
The DUO-E Study Design: Physician Perspectives on Key Features
Dr Shannon Westin
Professor of Gynecologic Oncology and Reproductive Medicine at The University of Texas MD Anderson Cancer Center
Dr Floor Backes
Professor, Division of Gynecologic Oncology and Director of Gynecologic Cancer Research at The Ohio State University

DUO-E Trial Design
DUO-E was a global, randomized, double-blind, placebo-controlled Phase III study
Exploring Adverse Events in DUO-E
Dr Shannon Westin
Professor of Gynecologic Oncology and Reproductive Medicine at The University of Texas MD Anderson Cancer Center
Dr Floor Backes
Professor, Division of Gynecologic Oncology and Director of Gynecologic Cancer Research at The Ohio State University

Safety in the dMMR Subgroup
Safety data for IMFINZI with CP are available for a total of 44 patients with advanced or recurrent dMMR endometrial cancer.
Patients received IMFINZI (1120 mg) with CP (every 3 weeks for up to six 21-day cycles) followed by IMFINZI (1500 mg every 4 weeks) or CP (every 3 weeks for up to six 21-day cycles) alone. Treatment was continued until disease progression or unacceptable toxicity.
The median duration of exposure to IMFINZI with CP was 14.8 months (range: 0.7 to 31.7).
Serious adverse reactions (ARs) occurred in 30% of patients who received IMFINZI with carboplatin and paclitaxel
The most common serious ARs defined as those occurring in 4% of patients or more were constipation and rash, which both occurred in 4.5% of patients
Permanent discontinuation of IMFINZI due to ARs occurred in 11% of patients
The AR that resulted in permanent discontinuation of IMFINZI in 4% or more of patients was rash at 4.5%
Dosage interruptions of IMFINZI due to ARs occurred in 52% of patients
-
ARs that required dosage interruptions of IMFINZI in 4% or more of patients were:
Anemia (11%)
Thrombocytopenia (9%)
Neutropenia (9%)
COVID-19 (9%)
Increased alanine transaminase (ALT) (4.5%)
Pneumonitis (4.5%)


Clinically relevant ARs in less than 10% of patients who received IMFINZI with carboplatin and paclitaxel included:
Autoimmune hemolytic anemia
Colitis
Immune-mediated thyroiditis
Infusion-related reaction
Interstitial lung disease
Myositis
Pneumonitis
Pulmonary embolism
Sepsis
Please see the IMFINZI Prescribing Information for further details.
IMFINZI + CP offers another option to treat patients with primary advanced or recurrent dMMR EC
IMFINZI in combination with carboplatin and paclitaxel followed by IMFINZI as a single agent is indicated for the treatment of adult patients with primary advanced or recurrent endometrial cancer that is mismatch repair deficient as determined by an FDA-approved test.
DUO-E Efficacy
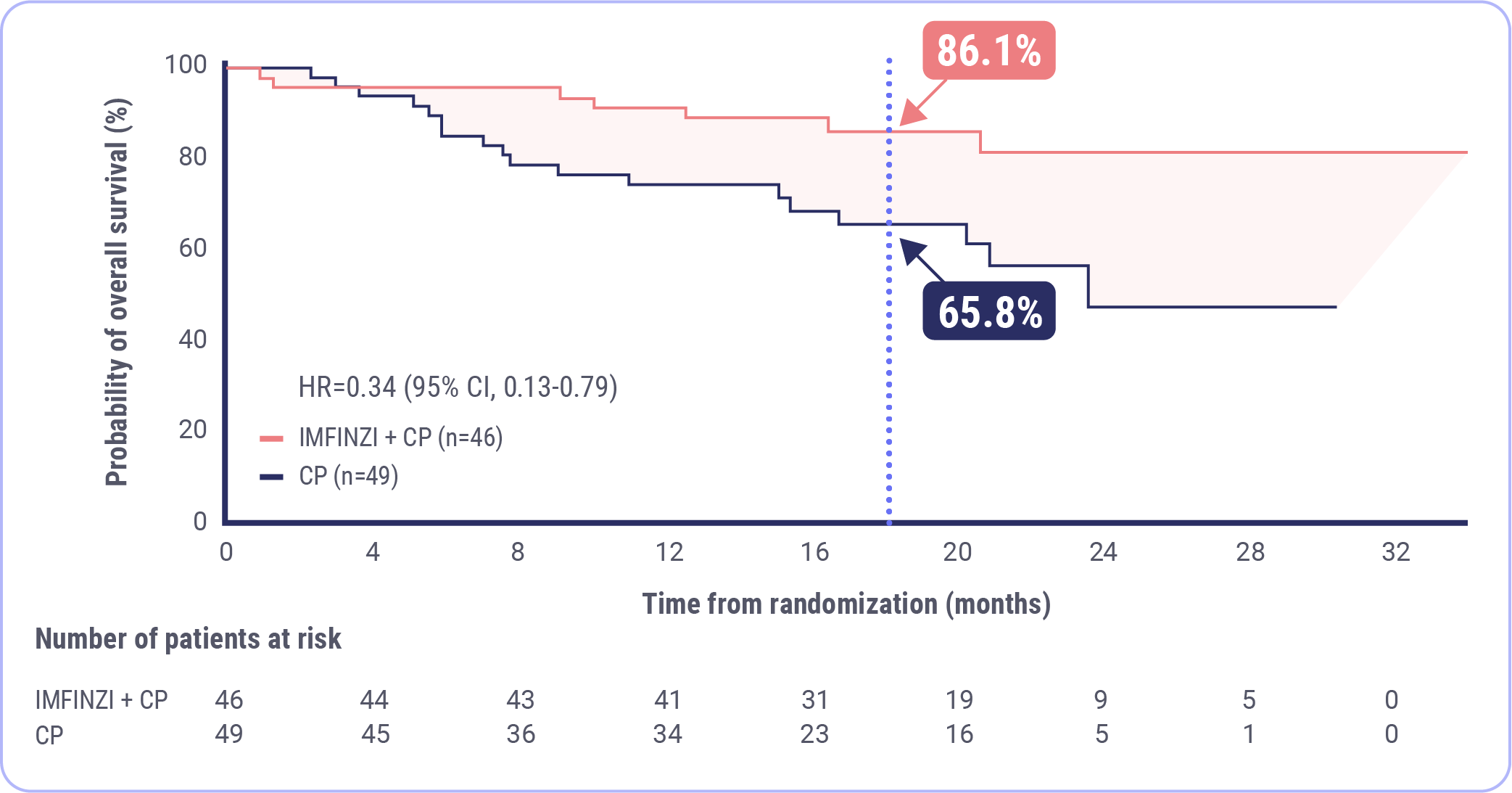
- The FDA approval of IMFINZI is supported by clinically meaningful data from DUO-E, which demonstrated that the combination of IMFINZI and CP reduced the risk of progression or death by more than half in patients with dMMR endometrial cancer (n=95). The prespecified PFS subgroup analysis was exploratory and not designed to assess a statistical difference between treatment groups
- IMFINZI + CP led to a 58% reduction in the risk of progression or death, with a hazard ratio of 0.42 (95% CI, 0.22-0.80)
- The median progression-free survival was not reached in the IMFINZI + CP arm (95% CI, NR-NR) and was 7 months in the CP arm (95% CI, 6.7-14.8)
- Testing patients to determine mismatch repair status at diagnosis is recommended to evaluate if patients are eligible to receive precision medicine treatments
- Select patients for treatment based on the presence of dMMR in tumor specimens. Information on FDA-approved tests for the detection of dMMR status in endometrial cancer is available at https://www.fda.gov/companiondiagnostics

DUO-E Study Design
In the DUO-E study, there are several trial design features that may influence results and interpretations. These features include:
- Geographic location diversity
- Inclusion of characteristics associated with aggressive disease
- The platinum-free interval for patients with recurrent disease
- Treatment duration
- The statistical analysis plan

DUO-E Safety
- Serious ARs occurred in 30% of patients who received IMFINZI + CP. The most common serious ARs (≥4%) were constipation (4.5%) and rash (4.5%)
- The most common adverse reactions (>20%), including laboratory abnormalities, were peripheral neuropathy, musculoskeletal pain, nausea, alopecia, fatigue, abdominal pain, constipation, rash, decreased magnesium, increased ALT, increased AST, diarrhea, vomiting, cough, decreased potassium, dyspnea, headache, increased alkaline phosphatase, and decreased appetite
- Permanent discontinuation of IMFINZI due to ARs occurred in 11% of patients. The AR that resulted in permanent discontinuation of IMFINZI in 4% or more of patients was rash at 4.5%
- Dosage interruptions of IMFINZI due to ARs occurred in 52% of patients. ARs that required dosage interruptions of IMFINZI (≥4%) were anemia (11%), thrombocytopenia (9%), neutropenia (9%), COVID-19 (9%), increased ALT (4.5%), and pneumonitis (4.5%)
IMFINZI in EC
The IMFINZI plus CP combination indication offers an additional treatment option for patients with primary advanced or recurrent dMMR endometrial cancer, thus expanding the treatment options.

Abbreviations
1L=first-line; ALT=alanine aminotransferase; ARs=adverse reactions; AST=aspartate aminotransferase; CI=confidence interval; CP=carboplatin and paclitaxel; dMMR=mismatch repair deficient; DoR=duration of response; EC=endometrial cancer; FDA=United States Food and Drug Administration; FIGO=International Federation of Gynecology and Obstetrics; HR=hazard ratio; ITT=intent to treat; MMR=mismatch repair; mOS=median overall survival; mPFS=median progression-free survival; NR=not reached; ORR=objective response rate; OS=overall survival; PFS=progression-free survival; Q3W=every 3 weeks; Q4W=every 4 weeks; RECIST=Response Evaluation Criteria in Solid Tumors.

References
American Cancer Society (ACS). Cancer Facts & Figures. 2025. Accessed June 2025. www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2025/2025-cancer-facts-and-figures-acs.pdf
American Cancer Society (ACS). About Endometrial Cancer. Accessed June 2025. https://www.cancer.org/cancer/types/endometrial-cancer/about/key-statistics.html
AstraZeneca. IMFINZI plus chemotherapy approved in the US for mismatch repair deficient advanced or recurrent endometrial cancer. Accessed June 2025. https://www.astrazeneca.com/media-centre/press-releases/2024/imfinzi-approved-in-the-us-for-endometrial-cancer.html
Baurain J-F, Chon HS, Thomes-Pepin J, et al. Durvalumab + carboplatin/paclitaxel followed by durvalumab +/- olaparib as a first-line treatment for endometrial cancer: overall survival and additional secondary efficacy endpoints by mismatch repair status in the DUO-E/GOG-3041/ENGOT-EN10 trial. Presented at: Society of Gynecologic Oncology Annual Meeting on Women’s Cancer; March 16-18, 2024; San Diego, CA.
Berg HF, Engerud H, Myrvold M, et al. Mismatch repair markers in preoperative and operative endometrial cancer samples; expression concordance and prognostic value. Br J Cancer. 2023;128(4):647-655.
Cao SY, Fan Y, Zhang YF, Ruan JY, Mu Y, Li JK. Recurrence and survival of patients with stage III endometrial cancer after radical surgery followed by adjuvant chemo- or chemoradiotherapy: a systematic review and meta-analysis. BMC Cancer. 2023;23(1):31.
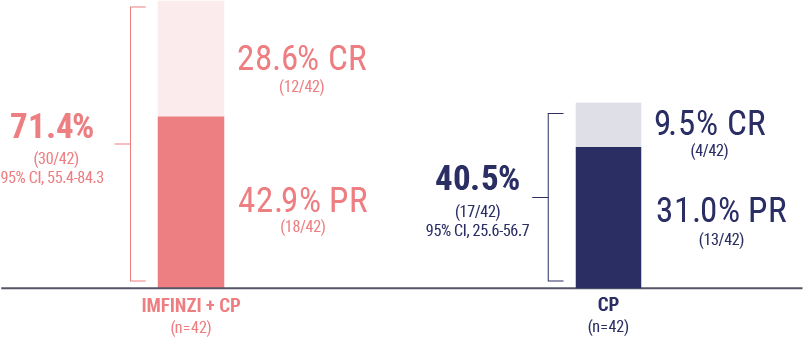
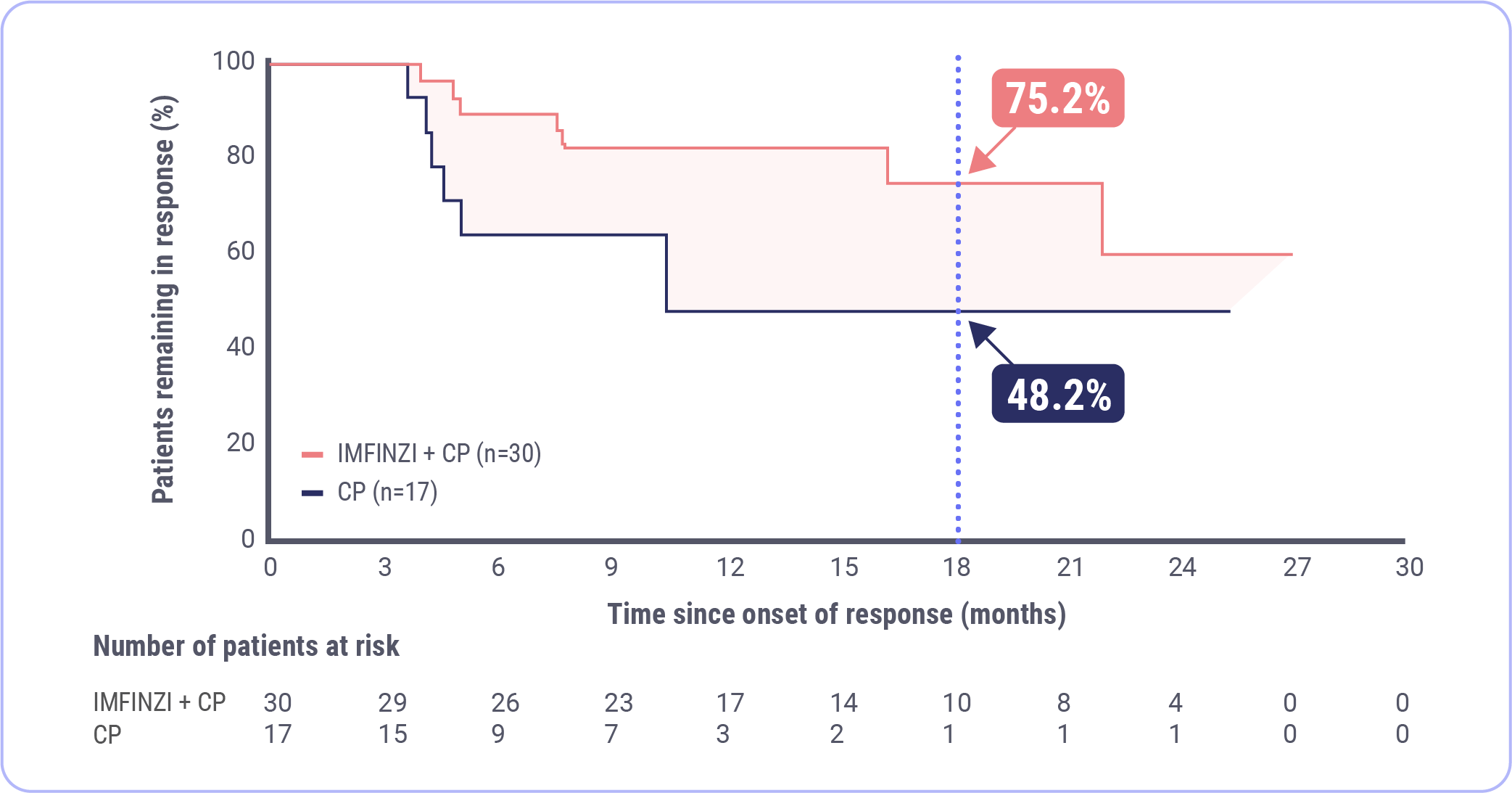
Chon HS, Thomes-Pepin J, Sundborg MJ, et al. Durvalumab + carboplatin/paclitaxel followed by durvalumab with or without olaparib as first-line treatment for endometrial cancer (DUO-E/GOG-3041/ENGOT-EN10): objective response rate and duration of response by mismatch repair status. Presented at: Society of Gynecologic Oncology Annual Meeting on Women’s Cancer; March 16-18, 2024; San Diego, CA.
Constantine GD, Kessler G, Graham S, Goldstein SR. Increased incidence of endometrial cancer following the Women’s Health Initiative: An assessment of risk factors. J Womens Health (Larchmt). 2019;28(2):237-243.
Data on File, REF-232005, AstraZeneca Pharmaceuticals LP; 2024.
Data on File, REF-233132, AstraZeneca Pharmaceuticals LP; 2024.
Hamoud BH, Sima RM, Vacaroiu IA, et al. The evolving landscape of immunotherapy in uterine cancer: A comprehensive review. Life (Basel). 2023;13(7):1502.
IMFINZI® (durvalumab) [Prescribing Information]. Wilmington, DE: AstraZeneca Pharmaceuticals LP; 2025.
Kim JJ, Kurita T, Bulun SE. Progesterone action in endometrial cancer, endometriosis, uterine fibroids, and breast cancer. Endocr Rev. 2013;34(1):130-162.
Li GM. Mechanisms and functions of DNA mismatch repair. Cell Res. 2008;18(1):85-98.
Li K, Luo H, Huang L, Luo H, Zhu X. Microsatellite instability: a review of what the oncologist should know. Cancer Cell Int. 2020;20:16.
Makker V, Green AK, Wenham RM, Mutch D, Davidson B, Miller DS. New therapies for advanced, recurrent, and metastatic endometrial cancers. Gynecol Oncol Res Pract. 2017;4:19.
Makker V, MacKay H, Ray-Coquard I, et al. Endometrial cancer. Nat Rev Dis Primers. 2021;7(1):88.
Monk BJ, Smith G, Lima J, et al. Real-world outcomes in patients with advanced endometrial cancer: A retrospective cohort study of US electronic health records. Gynecol Oncol. 2022;164(2):325-332.
National Cancer Institute (NCI). Cancer Stat Facts: Uterine Cancer. Accessed June 2025. https://seer.cancer.gov/statfacts/html/corp.html
Siegenthaler F, Lindemann K, Epstein E, et al. Time to first recurrence, pattern of recurrence, and survival after recurrence in endometrial cancer according to the molecular classification. Gynecol Oncol. 2022;165(2):230-238.
Tillmanns T, Masri A, Stewart C, et al. Advanced endometrial cancer-The next generation of treatment: A society of gynecologic oncology journal club clinical commentary. Gynecol Oncol Rep. 2024;55:101462.
Westin SN, Moore K, Chon HS, et al. Durvalumab plus carboplatin/paclitaxel followed by maintenance durvalumab with or without olaparib as first-line treatment for advanced endometrial cancer: The Phase III DUO-E Trial. J Clin Oncol. 2024;42(3):283-299.
Zhao P, Li L, Jiang X, Li Q. Mismatch repair deficiency/microsatellite instability-high as a predictor for anti-PD-1/PD-L1 immunotherapy efficacy. J Hematol Oncol. 2019;12(1):54.

For US Healthcare Providers only.
IMFINZI is a registered trademark of the AstraZeneca group of companies.
©2025 AstraZeneca. All rights reserved. US-107391 Last Updated 12/25

Important Safety Information
There are no contraindications for IMFINZI® (durvalumab).
Immune-Mediated Adverse Reactions
Important immune-mediated adverse reactions listed under Warnings and Precautions may not include all possible severe and fatal immune-mediated reactions. Immune-mediated adverse reactions, which may be severe or fatal, can occur in any organ system or tissue. Immune-mediated adverse reactions can occur at any time after starting treatment or after discontinuation. Monitor patients closely for symptoms and signs that may be clinical manifestations of underlying immune-mediated adverse reactions. Evaluate liver enzymes, creatinine, and thyroid function at baseline and periodically during treatment. In cases of suspected immune-mediated adverse reactions, initiate appropriate workup to exclude alternative etiologies, including infection. Institute medical management promptly, including specialty consultation as appropriate. Withhold or permanently discontinue IMFINZI depending on severity. See USPI Dosing and Administration for specific details. In general, if IMFINZI requires interruption or discontinuation, administer systemic corticosteroid therapy (1 mg to 2 mg/kg/day prednisone or equivalent) until improvement to Grade 1 or less. Upon improvement to Grade 1 or less, initiate corticosteroid taper and continue to taper over at least 1 month. Consider administration of other systemic immunosuppressants in patients whose immune-mediated adverse reactions are not controlled with corticosteroid therapy.
Immune-Mediated Pneumonitis
IMFINZI can cause immune-mediated pneumonitis. The incidence of pneumonitis is higher in patients who have received prior thoracic radiation.
Immune-Mediated Colitis
IMFINZI can cause immune-mediated colitis that is frequently associated with diarrhea. Cytomegalovirus (CMV) infection/reactivation has been reported in patients with corticosteroid-refractory immune-mediated colitis. In cases of corticosteroid-refractory colitis, consider repeating infectious workup to exclude alternative etiologies.
Immune-Mediated Hepatitis
IMFINZI can cause immune-mediated hepatitis.
Immune-Mediated Endocrinopathies
-
Adrenal Insufficiency: IMFINZI can cause primary or secondary adrenal insufficiency. For Grade 2 or higher adrenal insufficiency, initiate symptomatic treatment, including hormone replacement as clinically indicated.
-
Hypophysitis: IMFINZI can cause immune-mediated hypophysitis. Hypophysitis can present with acute symptoms associated with mass effect such as headache, photophobia, or visual field cuts. Hypophysitis can cause hypopituitarism. Initiate symptomatic treatment including hormone replacement as clinically indicated.
-
Thyroid Disorders (Thyroiditis, Hyperthyroidism, and Hypothyroidism): IMFINZI can cause immune-mediated thyroid disorders. Thyroiditis can present with or without endocrinopathy. Hypothyroidism can follow hyperthyroidism. Initiate hormone replacement therapy for hypothyroidism or institute medical management of hyperthyroidism as clinically indicated.
-
IMFINZI with Carboplatin and Paclitaxel
-
Immune-mediated hypothyroidism occurred in 14% (34/235) of patients receiving IMFINZI in combination with carboplatin and paclitaxel.
-
-
-
Type 1 Diabetes Mellitus, which can present with diabetic ketoacidosis: Monitor patients for hyperglycemia or other signs and symptoms of diabetes. Initiate treatment with insulin as clinically indicated.
Immune-Mediated Nephritis with Renal Dysfunction
IMFINZI can cause immune-mediated nephritis.
Immune-Mediated Dermatology Reactions
IMFINZI can cause immune-mediated rash or dermatitis. Exfoliative dermatitis, including Stevens-Johnson Syndrome (SJS), drug rash with eosinophilia and systemic symptoms (DRESS), and toxic epidermal necrolysis (TEN), has occurred with PD-1/L-1 antibodies. Topical emollients and/or topical corticosteroids may be adequate to treat mild to moderate non-exfoliative rashes.
Other Immune-Mediated Adverse Reactions
The following clinically significant, immune-mediated adverse reactions occurred at an incidence of less than 1% each in patients who received IMFINZI or were reported with the use of other PD-1/PD-L1 blocking antibodies.
-
Cardiac/vascular: Myocarditis, pericarditis, vasculitis.
-
Nervous system: Meningitis, encephalitis, myelitis and demyelination, myasthenic syndrome/myasthenia gravis (including exacerbation), Guillain-Barré syndrome, nerve paresis, autoimmune neuropathy.
-
Ocular: Uveitis, iritis, and other ocular inflammatory toxicities can occur. Some cases can be associated with retinal detachment. Various grades of visual impairment to include blindness can occur. If uveitis occurs in combination with other immune-mediated adverse reactions, consider a Vogt-Koyanagi-Harada-like syndrome, as this may require treatment with systemic steroids to reduce the risk of permanent vision loss.
-
Gastrointestinal: Pancreatitis including increases in serum amylase and lipase levels, gastritis, duodenitis.
-
Musculoskeletal and connective tissue disorders: Myositis/polymyositis, rhabdomyolysis and associated sequelae including renal failure, arthritis, polymyalgia rheumatic.
-
Endocrine: Hypoparathyroidism.
-
Other (hematologic/immune): Hemolytic anemia, aplastic anemia, hemophagocytic lymphohistiocytosis, systemic inflammatory response syndrome, histiocytic necrotizing lymphadenitis (Kikuchi lymphadenitis), sarcoidosis, immune thrombocytopenia, solid organ transplant rejection, other transplant (including corneal graft) rejection.
Infusion-Related Reactions
IMFINZI can cause severe or life-threatening infusion-related reactions. Monitor for signs and symptoms of infusion-related reactions. Interrupt, slow the rate of, or permanently discontinue IMFINZI based on the severity. See USPI Dosing and Administration for specific details. For Grade 1 or 2 infusion-related reactions, consider using pre-medications with subsequent doses.
Complications of Allogeneic HSCT after IMFINZI
Fatal and other serious complications can occur in patients who receive allogeneic hematopoietic stem cell transplantation (HSCT) before or after being treated with a PD-1/L-1 blocking antibody. Transplant-related complications include hyperacute graft-versus-host disease (GVHD), acute GVHD, chronic GVHD, hepatic veno-occlusive disease (VOD) after reduced intensity conditioning, and steroid-requiring febrile syndrome (without an identified infectious cause). These complications may occur despite intervening therapy between PD-1/L-1 blockade and allogeneic HSCT. Follow patients closely for evidence of transplant-related complications and intervene promptly. Consider the benefit versus risks of treatment with a PD-1/L-1 blocking antibody prior to or after an allogeneic HSCT.
Embryo-Fetal Toxicity
Based on their mechanism of action and data from animal studies, IMFINZI can cause fetal harm when administered to a pregnant woman. Advise pregnant women of the potential risk to a fetus. In females of reproductive potential, verify pregnancy status prior to initiating IMFINZI and advise them to use effective contraception during treatment with IMFINZI and for 3 months after the last dose of IMFINZI.
Lactation
There is no information regarding the presence of IMFINZI in human milk; however, because of the potential for serious adverse reactions in breastfed infants from IMFINZI, advise women not to breastfeed during treatment and for 3 months after the last dose.
Adverse Reactions
-
In patients with advanced or recurrent dMMR endometrial cancer in the DUO-E study receiving IMFINZI in combination with carboplatin and paclitaxel followed by IMFINZI as a single agent (n=44), the most common adverse reactions, including laboratory abnormalities (occurring in >20% of patients) were peripheral neuropathy (61%), musculoskeletal pain (59%), nausea (59%), alopecia (52%), fatigue (41%), abdominal pain (39%), constipation (39%), rash (39%), decreased magnesium (36%), increased ALT (32%), increased AST (30%), diarrhea (27%), vomiting (27%), cough (27%), decreased potassium (25%), dyspnea (25%), headache (23%), increased alkaline phosphatase (20%), and decreased appetite (18%). The most common Grade 3 or 4 adverse reactions (≥3%) were constipation (4.5%) and fatigue (4.5%).
-
In patients with advanced or recurrent dMMR endometrial cancer in the DUO-E study receiving IMFINZI in combination with carboplatin and paclitaxel followed by IMFINZI as a single agent (n=44), permanent discontinuation of IMFINZI due to adverse reactions occurred in 11% of patients. Serious adverse reactions occurred in 30% of patients who received IMFINZI with carboplatin and paclitaxel; the most common serious adverse reactions (≥4%) were constipation (4.5%) and rash (4.5%).
The safety and effectiveness of IMFINZI has not been established in pediatric patients.
Indication:
IMFINZI in combination with carboplatin and paclitaxel followed by IMFINZI as a single agent is indicated for the treatment of adult patients with primary advanced or recurrent endometrial cancer that is mismatch repair deficient (dMMR) as determined by an FDA-approved test.
Please see Full Prescribing Information including Medication Guide for IMFINZI.